The book, “Promoting Access to Medical Technologies and Innovation: Intersections between Public Health, Intellectual Property and Trade,” was launched on 5 February 2013, by the heads of the three bodies — the World Health Organization (WHO), World Intellectual Property Organization (WIPO) and World Trade Organization (WTO). Former Swiss President Ruth Dreifuss, who is also a former Chairperson of the World Health Organization’s Commission on Intellectual Property, Innovation and Public Health (CIPIH), chaired the event.
“Public health remains a clear imperative for the international community,” said Madame Dreifuss. “Promoting both medical innovation and access to the fruits of that innovation is indispensable for progress towards improved and more equitable health outcomes.
“But to achieve this result demands greater practical cooperation and dialogue within the international system — the launch of this study represents an important step forward in that direction.”
This study “demystifies an intricate and extremely complex landscape of laws and policies and makes them accessible to the non-specialist,” said WHO Director-General Dr Margaret Chan.
“In so doing, it sets out a comprehensive and coherent inventory of legal instruments and policy options that can be drawn on to craft measures that meet national public health objectives.”
WIPO Director General Francis Gurry said: “This joint WHO-WIPO-WTO publication will enhance our shared understanding of health, trade and intellectual property and provide policy makers with a comprehensive compendium of the issues at stake.
“We look forward to our continued collaboration, in particular to show that innovation and IP are essential components of an effective health policy.”
“Access to medicines requires the right mix of health policies, intellectual property rules and trade policy settings,” said WTO Director-General Pascal Lamy. “And coherence is key to finding sustainable solutions. This is the spirit behind the joint study: to provide well informed, comprehensive policy choices.”
Complex, linked issues
The book covers a broad range of complex, yet linked issues relating to public health and innovation in medical technologies, with the ultimate goal of accessibility — making medical advances available globally to all who are sick. It provides solid information for anyone concerned with these issues.
Projected global deaths from selected causes, 2004–30
Millions
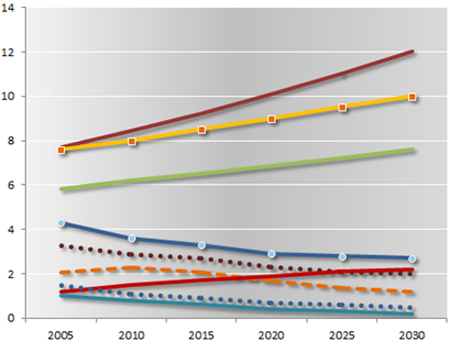
| Cancers | Ischaemic heart disease | Cerebrovascular disease |
| Acute respiratory infections | Perinatal conditions | HIV/ AIDS |
| Road traffic accidents | Tuberculosis | Malaria |
— page 27
Its target audiences are: policymakers, legislators, government officials, delegates to international organizations, non-governmental organizations, and researchers in this field.
The study reflects the debate about health that has evolved over the years, with increasing attention given to medical technologies and their invention and dissemination. Public health and innovation policies, and the rules of trade, competition and procurement, all play a part.
The policy-making focus has broadened from the basic questions of ensuring access to essential medicines, and developing treatments for neglected diseases that are available and affordable for those who are primarily affected — the poor. This is part of the right to health.
More recently, attention has turned to other aspects of how to meet this right: including the measures that are needed in order to provide incentives for medical innovations — such as medicines, vaccines and medical devices — and how to ensure equitable access to all of these vital medical technologies.
Part of the picture is the international patent system and how governments implement it domestically according to the needs of their countries. The patent system is designed to support innovation, and it offers a mechanism to ensure that these innovations are accessible to society.
International medical patent applications, 2000–2010
WIPO Patent Cooperation Treaty applications in medical technology, including pharmaceuticals
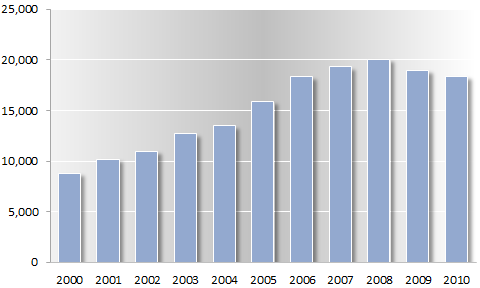
— page 63
The research-and-development pharmaceutical industry therefore relies heavily on exclusive patent rights in order to recoup the investment made in research and development, as shown by the high number of applications for patents on medical technologies under WIPO’s Patent Cooperation Treaty.
The secretariats of the three organizations have drawn on their experience and the data available to them to produce this study, and to support discussions on policy options and legal issues.
What the book covers
The book looks at the need for international cooperation, who is involved, and how to address the challenges that the sector is facing. It examines in detail the range of policy issues from health and human rights and national, regional and global regulation policies, to intellectual property, trade and tariffs, procurement, free trade agreements and other aspects of policy.
Import duties on pharmaceuticals and medical equipment
Trade-weighted average rates applied to health-related products
Developed countries Developing countries
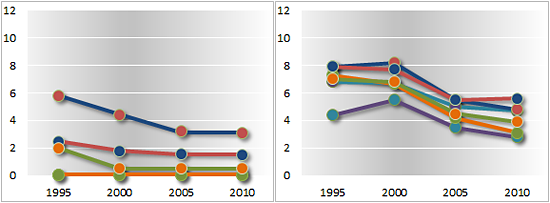
Least developed countries Transition economies
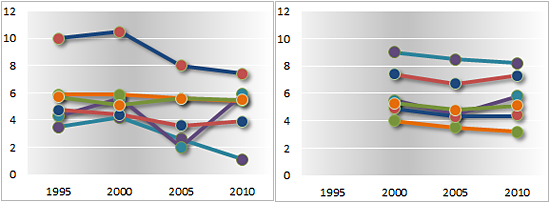
A1 = dosified medicines; A2 = bulk medicines; A3 = specific chemical inputs; B = general inputs; C1 = hospital inputs; C2 = medical equipment
— page 196
It studies a range of issues, such as: patents in the pharmaceutical sector; traditional medical knowledge; the importance of knowing what is patented and where, and how easy it is to find out; and questions of affordability and availability of medicines and market failure.
It looks in some depth at the development of medical technologies, modern research and development, ways of providing incentives for innovation, and ways of dealing with market failures, in particular with new products for treating neglected diseases. It includes comprehensive sections on trade and intellectual property rules and the flexibilities they contain for governments to meet various public health objectives.
More
- Two-page summary
- The study is available here: WHO website; WIPO website; www.wto.org/trilateralstudy2013
> Problems viewing this page?
Please contact [email protected] giving details of the operating system and web browser you are using.
![]() Audio:
Audio:
> Pascal Lamy’s introductory Remarks, Director-General, World Trade Organization (WTO)
> Mrs Dreifuss’s introductory Remarks, Chairperson
> Remarks: Pascal Lamy, Director-General, World Trade Organization (WTO)
> Remarks: Margaret Chan, Director-General, World Health Organization (WHO)
> Remarks: Francis Gurry, Director General, World Intellectual Property Organization (WIPO
> Pascal Lamy’s concluding remarks
“
WHO analysed availability and affordability of essential medicines in the public and private sectors in 46 [low to middle income countries] between 2001 and 2009. […] The global average median availability of such medicines in the public sector is less than 42%. Generally, availability of generics is higher in the private sector — almost 72% in the same studies — although in many parts of the world, the private sector prefers to stock originator products.”
— page 147



