One year of exporting COVID-19 vaccines: what does the evidence show?
While still unfolding, the COVID-19 vaccine experience is a story of light in terms of speed and reach. Just over two years after the first cases of SARS-CoV-2 were detected, around 10.5 billion vaccine doses have been administered globally. But it is also a story of shadows. Profound vaccine inequity continues to limit access in low-income countries (LICs) where, even today, only 11.4 percent of people have received at least one shot. With 4.4 billion doses having crossed borders as of December 2021, trade has proven instrumental to the manufacturing and distribution of vaccines across the world. The global trading system, underpinned by World Trade Organization (WTO) rules, has helped keep markets broadly open and supplies moving. Yet, greater trade cooperation in tackling geographic concentration, supply chain bottlenecks and regulatory divergences can strengthen preparedness for future pandemics.
A recent study on the historical record in the development and deployment of vaccines finds that never has the world seen such a rapid development of a viable vaccine, a massive ramp up of production and a fast and effective rollout of vaccination (Figure 1). Significant investments and public-private collaboration bolstered the emergence of the COVID-19 supply chain in the middle of the pandemic. While insufficient jabs significantly hampered vaccine access in 2021, supply-side constraints have begun to recede, and 24 billion doses are now forecast to be produced by mid-2022.
Figure 1. COVID-19 has been the fastest, most complex vaccine introduction globally in history.
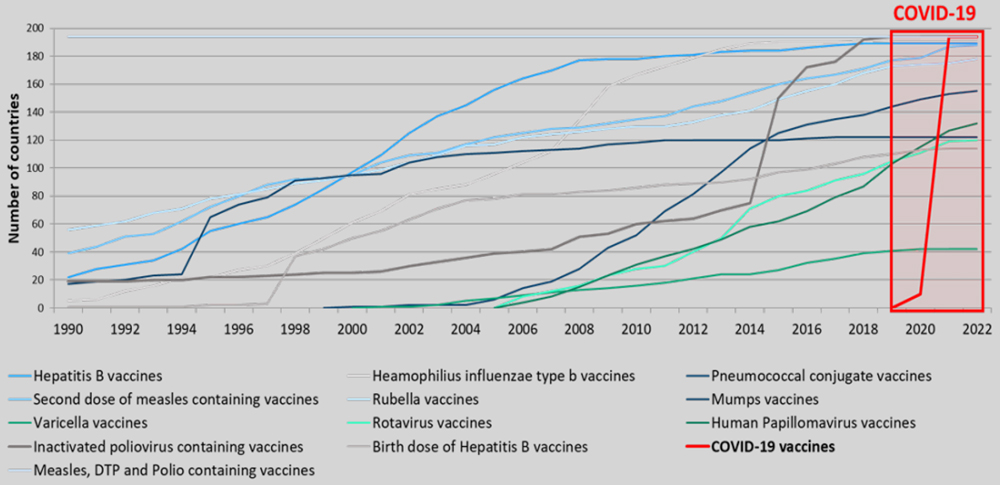
Source: Lindstrand (2022, February 11), Session 5: Getting Vaccines into Arms [Video of the WTO Technical Workshop on COVID-19 Vaccine R&D, Manufacturing and Distribution: How it is in Practice].
Despite the success in scaling up production, inoculation rates across countries at different stages of development are still unbalanced. The share of the population that has received at least one vaccine dose is almost 7 times higher in High-income countries (HICs) than in LICs. Africa continues to suffer from low levels of vaccination (Figure 2) and at least 20 countries in the region could miss a World Health Organization (WHO) target of dosing 70 percent of their populations by mid-year. While stockpiling of doses that could be redeployed in regions of greater need is still a risk, widespread country-specific challenges associated with delivery, demand and vaccine hesitancy, and cost and financing increasingly hinder diffusion of shots. Vaccine inequity is a global policy failure with negative consequences for the world at large. A recent paper argues that sharp disparities in allocation of vaccines between HICs and LICs lead to earlier and larger outbreaks of new waves; in contrast, equitable vaccine allocation strategies substantially curb the evolution and spread of new strains.
Figure 2. Share of people who received at least one vaccine dose (% of population)
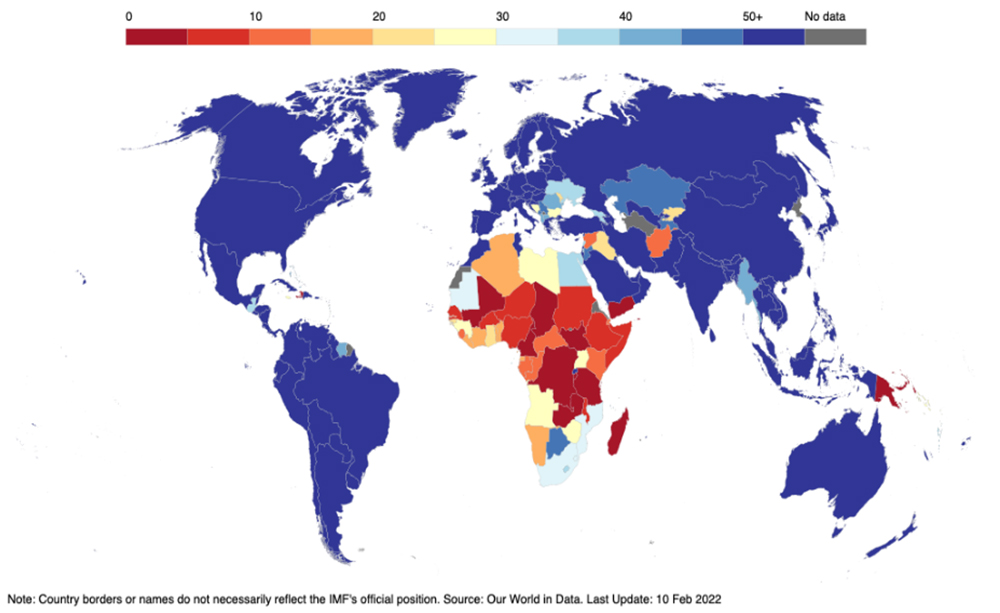
Source: IMF COVID-19 Global Targets and Progress Tracker.
Trade has been a force for good in fighting COVID-19. After a slow start, exacerbated by some trade policies, vaccine exports ramped up in the third quarter of 2021 to reach 4.4 billion doses by the end of the year (Figure 3). Such an outcome would not have been possible without the cross-border movement of dozens of specialized vaccine inputs along tightly knit supply chains. From vaccine core ingredients to vials and rubber stoppers to cold boxes, to consumables (such as specialized plastic bags), specialized machinery (such as bioreactors and cold chain equipment) and other products (such as dry ice and more), there is a high degree of trade interdependence between the ingredients and goods needed to produce, distribute, and administer vaccines. A stark lesson from 2021 is that trade is an indispensable conduit to vaccine access.
Figure 3. Billion COVID-19 vaccine doses exported by producing economy (cumulative)
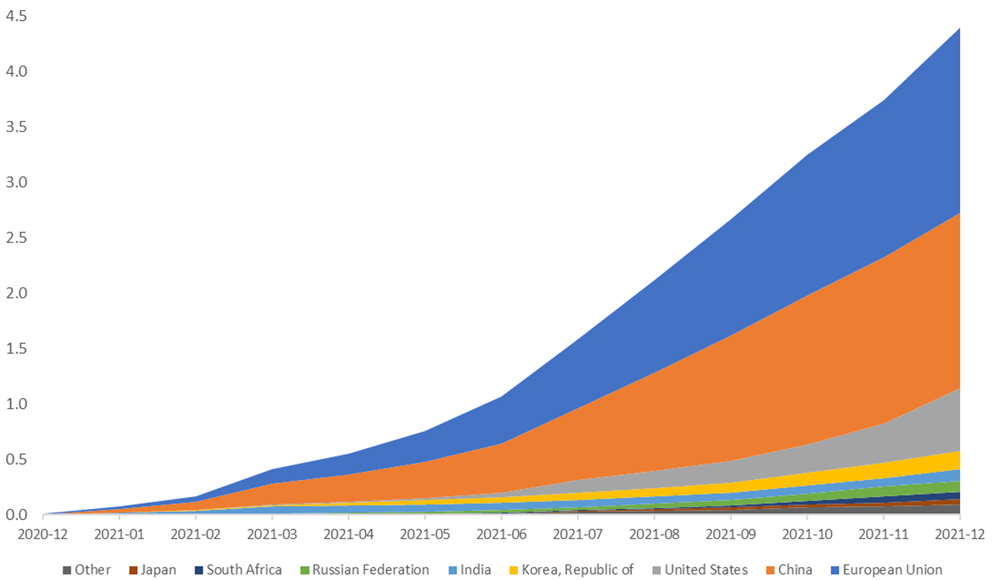
Source: WTO-IMF COVID-19 Vaccine Trade Tracker .
For all its central role in immunizing the world, the 2021 export experience has revealed challenges and shortcomings of vaccine trade. First, vaccine production is highly concentrated, with 87 percent of the total number of doses exported last year supplied by the European Union, China and the United States. While manufacturing under licensing agreements has played an important role in serving some markets, the main ingredient of vaccines — the drug substance — is produced in only nine economies (counting the European Union as one) (Figure 4). As governments prioritize their own populations, the fallout of geographic concentration is often limited exports of shots in moments of crisis, at least until production ramps up. To help manage the risk of vaccine inequity, supporting sustainable manufacturing capacities in all regions, Africa in particular, is a key priority. WHO recently announced that Egypt, Kenya, Nigeria, Senegal, South Africa and Tunisia will receive the technology needed to make mRNA vaccines in the continent, which will help tackle ongoing and future health crises.
Figure 4. COVID-19 drug substance capacity production by location
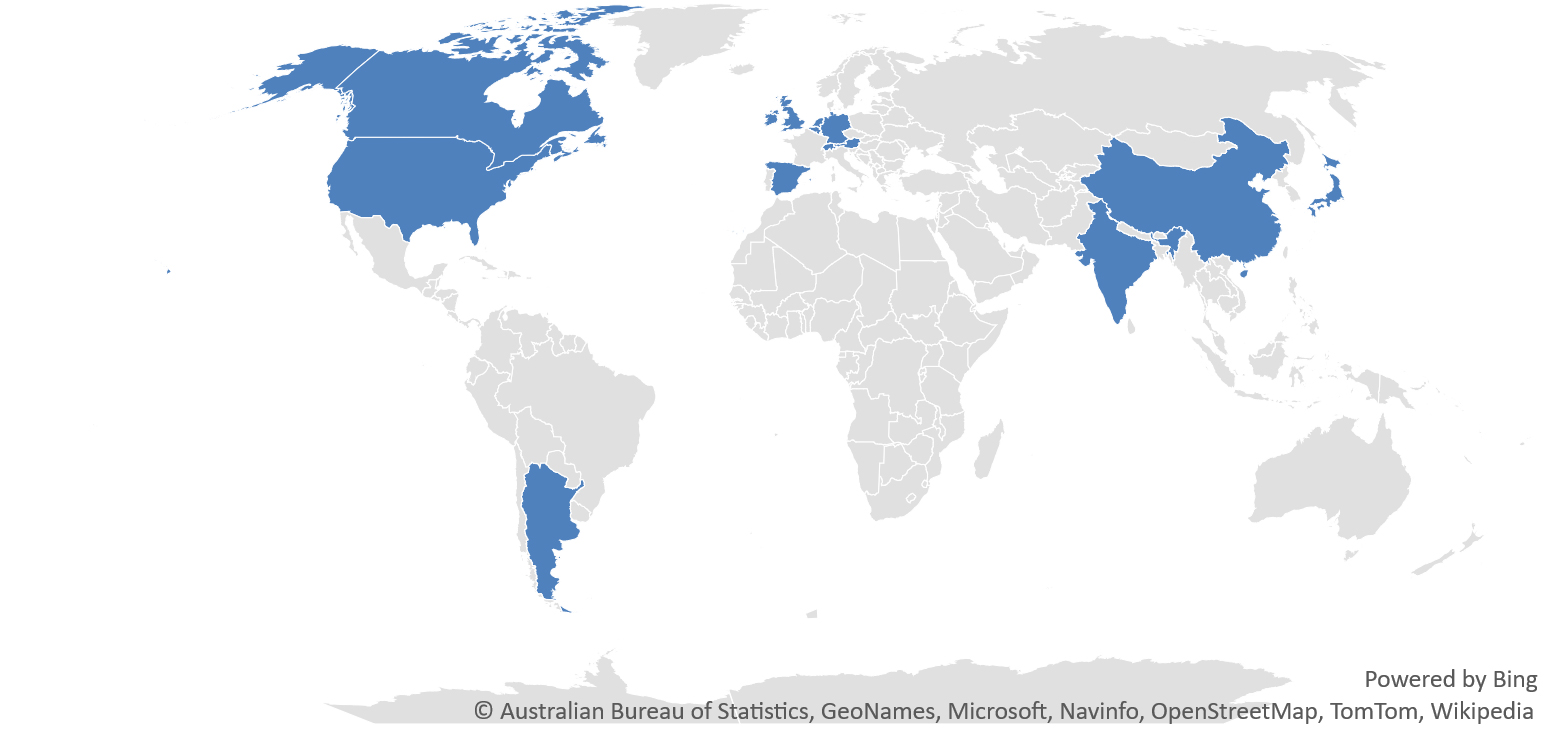
Source: COVID-19 Vaccine Market Dashboard | UNICEF Supply Division
Governments have taken steps to speed up and expand trade in many COVID-19 goods including by eliminating tariffs, smoothing or digitizing customs procedures and introducing green lanes for medical products and their inputs. But trade restrictions and logistics bottlenecks have exacerbated and magnified the pitfalls of export supply concentration. Vaccine manufacturers reported that export curbs have affected access to vaccine inputs as well as exports of finished vaccines and even the conduct of clinical trials. Some of these restrictions have been lifted, for example in the European Union and in India, easing the flow of vaccine doses and vaccine inputs across countries, but others are still in place.
Vaccine regulatory divergence has proven to be one major hurdle in the fight against COVID-19. While the WHO Emergency Use Listing Procedure has provided governments with a set of available quality, safety, and efficacy and performance data to facilitate emergency authorization of vaccines, different regulatory frameworks, procedures and timelines have added complexity and delayed delivery of jabs. Manufacturers still confront additional in-country requirements, unique labelling and other specifications, duplicative procedures and other regulatory barriers to expanding and optimizing production post-approval. Paperwork often needs to be resubmitted when a new production facility is opened or a production line is modified, which interrupts supply chains and discourages expansion of investments into new locations.
More recently, attention has moved to country readiness and absorption capacity for vaccines once they are received, with the Africa Centres for Disease Control and Prevention requesting that donations be paused until later in the year. While different measures are required to address these challenges, trade can be part of the answer by facilitating access to ancillary vaccine supplies such as syringes, needles, saline, personal protective equipment kits and safe waste-disposal mechanisms, as well as equipment needed to increase cold chain storage capacity. Timely access to such goods can prevent costly delays or potential wastage of doses, particularly given their short shelf life. As vaccination expands in many countries, protection against the risk of illicit vaccine trade is also important.
In the global race to vaccination, policies to support production and equity are needed. The Multilateral Leaders Task Force, integrated by the heads of the WTO, WHO, the World Bank and the International Monetary Fund has been backing countries across the world in their efforts to vaccinate their populations. But trade policy cooperation is a critical part of the solution to end vaccine inequity and fast-track delivery. Governments are currently at work, discussing their collective response as WTO Members to the pandemic, including on the controversial issue of the role of intellectual property rights in vaccines. The global trading system played a key role in helping the world move COVID-19 vaccines from the lab, out the factory gate and into people's arms, at record speed and massive scale. But insights gained from this pandemic must feed into our efforts to enhance international cooperation for better future pandemic preparedness.
* Anabel González was Deputy Director-General from June 2021 to August 2023
